Bring Your Own CRO
May 7, 2026
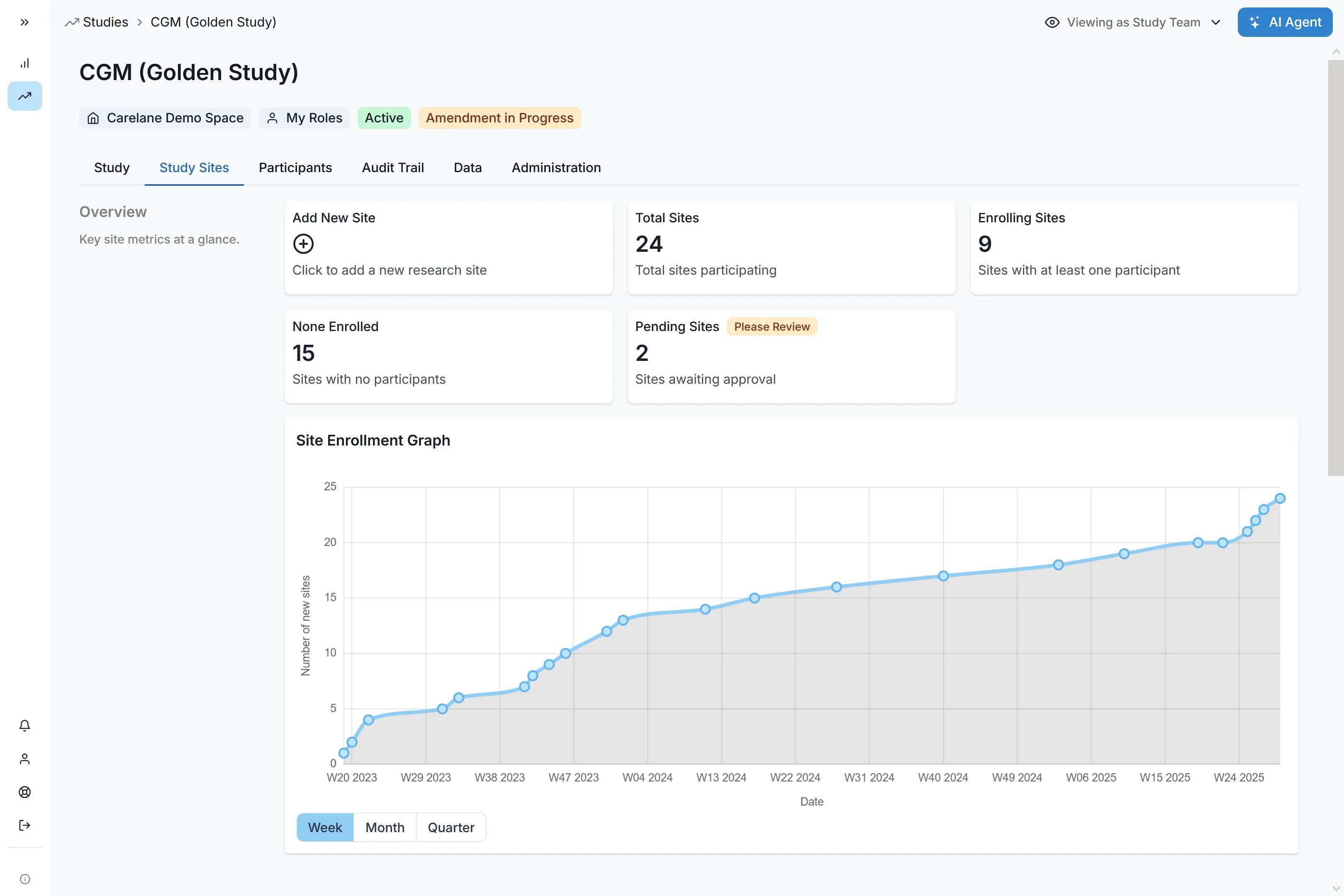
You Don't Own Your Trials. You Should.
Why the next decade will be defined by the teams who own their clinical infrastructure - and how "Bring Your Own CRO" becomes the new default.
In our last piece on the velocity paradox, we made the case that speed is no longer the enemy of quality - it's the ultimate risk mitigation strategy. Nowhere is that more true than in biotechnology, where every month of operational drag is a month of runway burned and equity diluted.
But for biotechs, there's a deeper problem hiding underneath the velocity question. One that no amount of "better project management" will fix.
You don't actually own your trials.
The Inconvenient Truth About Your eClinical Stack
Big pharma figured this out years ago. Walk into Pfizer or Roche and the eClinical stack - Veeva, Medidata, Oracle, whatever the vendor - is hosted by the vendor but owned by the sponsor: their licence, their configuration, their data, their admin rights. Vendor change does not equal study change. None of this is new at the top of the industry.
What is new is that the same arrangement is finally available to small biotechs at a footprint that doesn't require a 30-person clinical IT function. Until very recently, that wasn't true - and it's why almost every emerging biotech today is still operating under the old default.
Walk into almost any emerging biotech today and ask a simple question: "Who controls the systems running your Phase 2?"
The honest answer, in nearly every case, is: not you.
eClinical systems - EDC, CTMS, eTMF, the works - were never built with biotechs in mind. They were sold to CROs. The CRO buys the licence. The CRO configures the build. The CRO holds the admin keys. As the biotech footing the bill, you get seats. They get the keys.
That single architectural decision, made decades ago, when clinical research was effectively synonymous with big pharma and biotechs barely existed as a category, leads into every operational headache you experience today:
- Your data lives on their servers. Your team works off extracts, on someone else's refresh cadence. The number you need to brief your board on Tuesday sits in a database you can't query directly.
- Your CRO is unswappable. Switching CROs mid-programme means switching systems mid-study. Underperformers stay because moving costs more than tolerating them.
- Your protocol is frozen by your tooling. Every amendment becomes a change order on the vendor's timeline. The science waits for the ticket queue.
- Your portfolio is a patchwork. Each new study brings a new CRO, a new stack, a new set of templates. Nothing compounds.
You are, in effect, renting access to your own clinical programme. And you're paying full price for it.
The Ownership Flip
The fix isn't a better CRO. It isn't a faster EDC. It’s extending what big pharma already takes for granted to the rest of the industry.
%20(1).png)
The Ownership Flip: same data, same standards, different power structure.
We call the new model Bring Your Own CRO. The mental shift is simple, but it changes everything downstream:
- Today: the platform belongs to your CRO. Switch CROs, switch systems.
- Tomorrow: the platform belongs to you. The CRO logs into your infrastructure, does great work, and stays swappable.
Your data lives in your environment, in open formats. Your protocol library, your edit checks, your audit trail — all yours. The CRO brings expertise and execution. You bring the spine that everything runs on.
Same data. Same compliance standards. Different power structure.
What Ownership Actually Gets You
This isn't a philosophical preference. The economics of biotech-owned infrastructure land directly on the four metrics every biotech board cares about: runway, valuation, agility, and time-to-readout.
1. Six months of runway. Per trial.
Build, amendments, and database lock typically eat ten months of calendar time across a study. When the protocol library, edit checks, and form templates are yours and reusable, that compresses to under four. That's six months of runway returned per trial — across a pipeline of three or four studies, you're talking about years.
For a biotech, those years are the difference between negotiating your Series B from strength and accepting a dilutive down-round to keep the lights on. As we said in the last piece: speed is equity preservation.
2. One spine across the entire portfolio.
When every study lives on the same platform you own, templates and patterns compound. Trial #7 is significantly cheaper to stand up than trial #1 — same backbone, same playbook, same trained team. Today, with each study on a different CRO's stack, you start from zero every time. Nothing compounds. Every win is a one-off.
3. A genuinely swappable CRO.
When the data and the infrastructure stay with you, the CRO becomes what they were always meant to be: an execution partner you can hire, evaluate, and replace on the merits. You change the team without changing the trial. Performance conversations stop being existential threats and start being normal vendor management.
This single change quietly resets the entire biotech–CRO power dynamic.
4. A readout you can actually time.
No more waiting on the Friday extract. No more reconciling three versions of "the latest enrolment number." You're briefing the board on this week's data, from live source. You amend the protocol when the science demands it, not when the vendor opens a ticket.
In a market where biotechs live and die on the cadence of their data readouts, owning that cadence is a strategic asset, not a nice-to-have.
"Stop renting access to your own science. The biotechs that win the next decade will be the ones that own their stack."— Yannick Boerner
But Isn't Owning the Stack a Massive Lift?
This is the question that has kept the old model in place for fifteen years. And it's a fair one. Historically, "own your stack" meant standing up an in-house clinical IT function: validating EDC, hiring CTMS admins, managing infrastructure. For a 30-person biotech, that's clearly absurd.
That's why Carelane exists.
Carelane is built specifically so that owning your platform doesn't require owning an IT department. You get an institutional-grade Clinical Trial Operating System out of the box, with the agility a modern biotech actually needs:
- Study-in-a-Box simplicity. A team of ten operates with the capability of a team of fifty. Whether you're running a multi-centre IIT or a Phase 2, the end-to-end setup is one platform, one login, one source of truth.
- Zero-friction onboarding. Site staff, nurses, and PIs can use the system with virtually no training. The "onboarding lag" that stalls study starts simply isn't there.
- Compliance by design. FHIR-based structured protocols, immutable audit trails on every action (including AI-assisted ones), zero-knowledge encryption, and strict role-based access control. Architectural data isolation means your data is never used to train external models. Innovation without audit anxiety.
- CRO-friendly by default. Your CRO logs into your platform and works the way they already work. No retraining, no friction — just a clearer line between who runs the trial and who owns it.
Buy for the Size You Intend to Be.
There's one operational caveat worth naming, because it's where most of the regret in this category actually lives. The platform you pick for your first IND has to still work when you have three programmes running across two continents and a new CRO partner who has never seen your stack before. The worst possible time to re-platform is around the pivotal trial — which is also exactly when teams who chose for today, rather than tomorrow, find out their tooling can't go where the science needs it to.
The decision isn't philosophical. It's logistical. Consider for the size you are. Buy for the size you intend to be.
The Decade Ahead
For most of the last twenty years, biotechs have accepted an inverted power structure their big-pharma counterparts never lived with: you take the scientific risk, raise the capital, and pay the bills, while the CRO holds the keys to the infrastructure that runs the science.
That arrangement made sense when the alternative was building your own clinical IT stack from scratch. It doesn't make sense anymore.
The biotechs that compound advantages over the next decade, the ones who get to Series B faster, fail bad assets sooner, and get good ones to readout while they still have runway, will be the ones that took back ownership of their trial infrastructure early.
You raised the money.
You took the scientific risk.
You should hold the keys.
Ready to try Carelane?
The new standard for Sponsors, CROs and Institutions to run faster trials. Instant adoption. Human-in-the-loop AI. Zero compliance risk.